The goal of the Molecular Physiology and Toxicology Laboratory (MPTL) is to mitigate the impact arthropod pests have on public health, animal health, and agriculture.
Physiology and Toxicology of G-Protein-Coupled Receptors
|
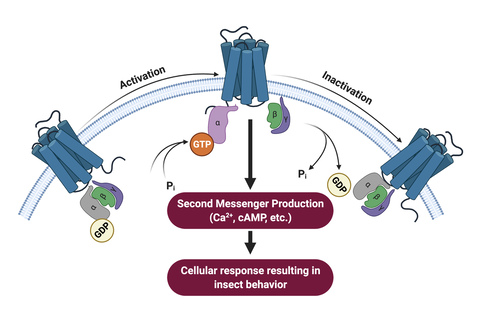
G-Protein-Coupled Receptors (GPCRs) are ubiquitously expressed in arthropod tissues and participate in several physiological processes. Activation of these receptors stimulate cellular signal transduction pathways that alter tissue function and/or behavior. The MPTL investigates the physiological relevance of GPCRs and GPCR-related pathways as potential insecticide targets. Approaches used to study GPCRs and related pathways include biochemical assays, electrophysiology, gene drive and genome editing technology, and heterologous expression. |
Insecticide and Acaricide Resistance
|
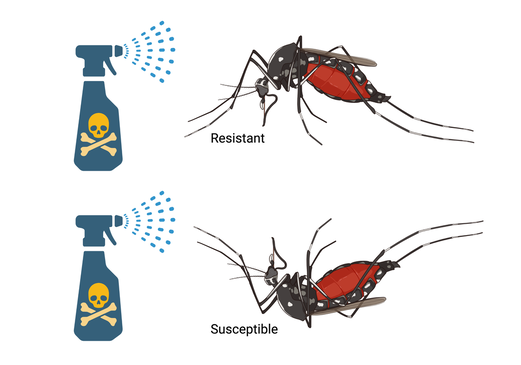
Widespread use and over reliance on pesticides has resulted in arthropods developing mechanisms of resistance. The MPTL seeks to identify and understand how target site mutations affect the physiology, pharmacology, and toxicology of affected proteins/ enzymes. Research includes understanding the physiological adaptations that allows for success of resistant populations. In addition to target-site mutations, we investigate expression and/or biochemical changes of target proteins/enzymes. Resistance studies are performed in arthropods that affect urban environments, agricultural pests, but also mosquitoes and ticks that affect public and/or animal health, along with model organisms.
|
Pollinator Health: Control of Honey Bee Pests and Parasites
|
Varroa destructor (Varroa Mites)
Mites are a devastating parasite to the western honey bee and a vector of several honey bee viruses. Mite populations in the hive are commonly controlled with the use of chemical acaricides. MPTL research has focused on measuring the acaricide resistance in different geographical regions of Virginia along with biochemical mechanisms that are involved in the metabolism of acaricides. Small hive beetles (Aethina tumida) Small hive beetles are ferocious feeders of honey bee larvae and colony products. An established laboratory colony is used for the development of toxicity bioassays using different xenobiotic exposure routes. Research has also focused on the use of novel acetylcholinesterase inhibitors that display a low level of non-target toxicity to mammals and honey bees. We are also exploring the olfactory system to pull beetles from an apiary and push them towards traps. Finally, we are investigating the relationship between small hive beetles and the fungus, Kodamaea ohmeri. |
|
Tick-host Interactions
|
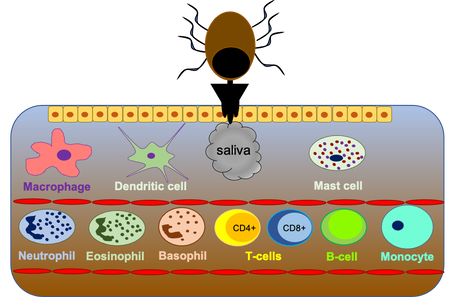
Tick saliva is known to contain a variety of pharmacologically active components that are involved in modulating the host’s immune response, but are also involved in salivary-assisted transmission of tick pathogens. We focus on the ability of ticks to modulate the non-neuronal cholinergic system of their host allowing ticks to feed for long periods of time undetected. Additionally, we are interested in understanding the mechanisms that are involved in the development of red meat allergies associated with tick bites.
|